
“A Clinically proven Herbal Composition to reduce viral load in Covid 19 Infection”
Keywords: COVID19 , CORONA , RTPCR . VIRAL LOAD, BLACK FUNGUS, MUCORMYCOSIS, ENVELOPE NON ENVELOP VIRUS, INFLUENZA , PCR ,RT-qPCR
Abstract
Herbal composition shows remarkable reduction in viral load more than in patients suffering from Covid 19 positive in to 4 days. Pre and post treatment viral load detection done by Quantitative reverse transcription PCR (RT-qPCR).
Invitro studies indicates activity against Envelop and Non-envelop virus category MS2 &INFLUENZA respectively. Antifungal activity against one of species responsible for mucor mycosis.
Pharmacodynamics – Significantly reduces viral load in 3 to 4 days. As per molecular docking studies, phytochemicals like Piperine, , Zingiberene are antiviral . They are having activity against covid spyke glycoprotein. as well as ACE II receptor blocker so works as a prophylaxis.
Clinical trial – In Vivo Human – CTRI/2021/03/032471
STUDY TITLE – “A Non-Interventional, Retrospective, Observational study to analyze safety, efficacy and tolerability of THINQURE 20 in COVID-19 patients.”
Study Design – Single arm retrospective study of 30 patients diagnosed as Covid19 positive with mild to moderate symptoms. Objective to access decline in reduction of % Viral load in 96 hrs RTqPCR (quantification of viral load ) method
Outcomes
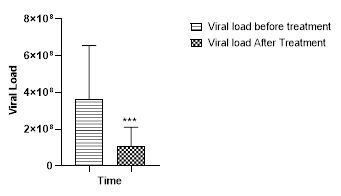
The treatment with Thinqure 20 for 5 days significantly (p<0.001) reduced the viral load in the Covid 19 Patients when compared with viral load before treatment
Non Clinical Study – In Vitro (Antiviral & Antifungal)- Study conducted in Mumbai NABL & GLP certified laboratory.
Outcomes
.
Thinqure20 – Safety and worldwide acceptance of herbal composition
Published Papers –
PATENT: – Primary specifications filed as a “HERBAL COMPOSITION FOR COVID 19 INFECTION”
SUMMARY:-THINQURE20 is a herbal composition influences by Ayurvedic texts showing excellent antiviral activity against Covid and other respiratory virus infections. Also showing activity against one of the species responsible for mucormycosis.

A product of THINQ Pharma-CRO Limited
Copyright © 2020 THINQ Pharma-CRO Limited All rights reserved
Disclaimer: Please note that the information provided is for the THINQURE 20 product information . Refer to your local government and/or health organizations for the most accurate and up to date information.